Understanding This Changed How I Think About Air, Not Just Chemistry
For a long time, I thought of CO₂ as something temporary.
We breathe it out.
Plants absorb it.
Ventilation removes it.
So in my mind, CO₂ felt fleeting — like something that comes and goes easily.
That assumption turned out to be wrong.
What I eventually learned is this:
👉 Carbon dioxide (CO₂) is a remarkably stable molecule — and that stability explains almost everything about how it behaves in real environments.
Once I understood that, a lot of questions suddenly made sense.
What “Stable” Really Means in Chemistry
When chemists say a molecule is stable, they don’t mean:
- harmless
- inactive
- unimportant
They mean something very specific:
👉 The molecule does not easily break apart, react, or transform under normal conditions.
CO₂ is stable because:
- its atoms are strongly bonded
- its structure is energetically favorable
- it requires significant energy to change
That stability defines how CO₂ behaves in air.
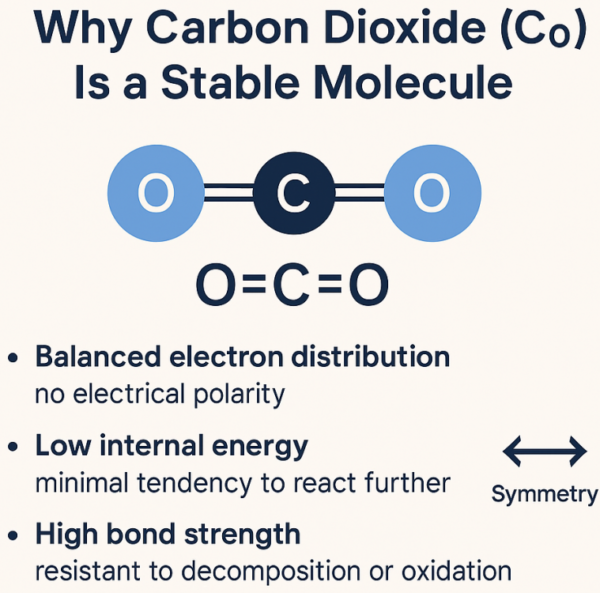
The Molecular Structure That Makes CO₂ Stable
At a molecular level, CO₂ is simple — but elegant.
It consists of:
- one carbon atom
- two oxygen atoms
Arranged in a straight line:
O = C = O
Those double bonds are strong.
They:
- lock the atoms in place
- lower the molecule’s energy
- make spontaneous reactions unlikely
Under everyday conditions — room temperature, normal pressure — CO₂ just exists.
It doesn’t decay.
It doesn’t react.
It doesn’t disappear.
Why CO₂ Doesn’t “Break Down” in Air
This was a key realization for me.
Many people intuitively assume:
“If CO₂ builds up, won’t it eventually dissipate or neutralize itself?”
But chemistry doesn’t work that way.
Because CO₂ is stable:
- it doesn’t decompose on its own
- it doesn’t react with oxygen or nitrogen
- it doesn’t get filtered out by most materials
Unless something actively removes or converts it, CO₂ simply accumulates.
That “something” can be:
- ventilation (air exchange)
- photosynthesis (in plants)
- industrial chemical processes
But not time alone.
Why Air Purifiers Don’t Remove CO₂
Understanding molecular stability made this obvious in hindsight.
Air purifiers are designed to:
- trap particles
- adsorb reactive gases
- capture large or polar molecules
CO₂ is:
- small
- non-reactive
- chemically satisfied
So it passes straight through most filters.
It’s not because purifiers are bad —
it’s because CO₂ doesn’t want to stick to anything.
Its stability makes it slippery.
Stability Is Why CO₂ Accumulates Indoors
This is where chemistry meets daily life.
In enclosed spaces:
- cars
- bedrooms
- offices
- RVs
CO₂ is constantly added by breathing.
Because it’s stable:
- it doesn’t decay
- it doesn’t neutralize
- it doesn’t get “used up”
So concentration rises until fresh air replaces it.
The molecule’s stability turns small continuous inputs into large accumulated effects.
Stability ≠ Danger — But Stability ≠ Irrelevance
This distinction matters.
CO₂ is stable, but that doesn’t mean:
- it’s toxic at normal levels
- it’s dangerous in small amounts
At the same time, stability means:
- it persists
- it accumulates
- its effects are cumulative
CO₂ doesn’t cause alarm.
It causes gradual change.
And gradual change is exactly what humans are worst at noticing.
Why Nature Treats CO₂ Differently
One reason CO₂ feels “natural” is because nature knows how to handle it.
Plants use energy from sunlight to:
- break CO₂’s stable bonds
- convert it into sugars
But that process:
- requires energy
- takes time
- only happens under the right conditions
Inside a car or room, that pathway doesn’t exist.
So CO₂ stays as it is.
The Shift in How I Think About CO₂ Now
I no longer think of CO₂ as something that:
“Goes away if I wait.”
I think of it as something that:
Only changes when I actively change the air.
That’s the practical implication of molecular stability.
Ventilation isn’t optional.
It’s the only mechanism that works in everyday spaces.
Final Thoughts
CO₂ is stable because:
- its molecular bonds are strong
- its structure is energetically efficient
- it doesn’t react under normal conditions
That stability is why:
- it accumulates
- it resists filtration
- it requires ventilation
- it quietly shapes indoor air quality
Once I understood that CO₂ is stable by design, not by accident,
I stopped expecting it to behave like smells, dust, or pollutants.
It’s not noisy.
It’s not dramatic.
It’s persistent.
And understanding persistence changes how you manage air — calmly, intentionally, and without misunderstanding what chemistry is actually doing.
Amazon is a trademark of Amazon.com, Inc. or its affiliates.
Leave a Reply